Sea ice
Contents
- 1 Nature of Sea Ice
- 2 Importance of Sea Ice
- 3 Holocene Sea Ice
- 4 Recent Trends in Sea Ice Growth
- 5 What is the difference between sea ice and icebergs, glaciers, and lake ice?
- 6 Characteristics
- 7 Processes
- 8 See Also
- 9 References
- 10 Citation
Nature of Sea Ice
Sea ice is simply frozen ocean water. It forms, grows, and melts in the ocean. In contrast, icebergs, glaciers, ice sheets, and ice shelves all originate on land. When sea ice melts, there is no effect on sea level, due to the density difference in seawater and sea ice. Thus a rise in ocean temperature that leads to sea ice melt has no effect on worldwide sea levels. Sea ice occurs in both the Arctic and Antarctic regions. In the Northern Hemisphere, it can currently exist as far south as Bohai Bay, China (approximately 38 degrees north latitude), which is actually about 700 kilometers (km) (435 miles) closer to the Equator than it is to the North Pole. In the Southern Hemisphere, sea ice only develops around Antarctica, occurring as far north as 55 degrees south latitude.
Sea ice generally grows during the winter months and melts during the summer months, but some sea ice remains all year in certain regions. About 15 percent of the world's oceans are covered by sea ice during part of the year. Recent studies in the Orkney Islands show that sea ice production has been expanding in the austral summer months. (Meredith et al, 2011) When sea ice melts or forms, there is no change in world sea level, due to the density difference in ice and seawater.
Importance of Sea Ice
Even though sea ice occurs primarily in the polar regions, it influences the global climate. Sea ice has a bright surface, so much of the sunlight (Solar radiation) that strikes it is reflected back into space. As a result, areas covered by sea ice don't absorb much solar energy, so temperatures in the polar regions remain relatively cool. If gradually warming temperatures melt sea ice over time, fewer bright surfaces are available to reflect sunlight back into space, more solar energy is absorbed at the surface, and temperatures rise. This chain of events can start a cycle of warming and melting. This cycle is halted when the dark days of the polar winter return, but it starts again in the following spring.

Sea ice also affects the movement of ocean waters. When sea ice forms, most of the salt is pushed into the ocean water below the ice, although some salt may become trapped in small pockets between ice crystals. Water below sea ice has a higher concentration of salt and is more dense than surrounding ocean water, and so it sinks. In this way, sea ice contributes to the ocean's global "conveyor-belt" circulation. Cold, dense, polar water sinks and moves along the ocean bottom toward the equator, while warm water from mid-depth to the surface travels from the equator toward the poles. Changes in the amount of sea ice can disrupt ocean circulation, thereby leading to changes in global weather.
Too much or too little sea ice can be a problem for wildlife and people who hunt and travel in polar regions. In the Arctic, sea ice can be an obstacle to normal shipping routes through the Northern Sea route and Northwest Passage.
Holocene Sea Ice
During the early Holocene, terrestrial Earth temperatures increased dramatically, melting much of the glaciers and sea ice on Earth. Over the period 12000 to 7000 years before present, the sea ice attained a level far below that of 2021, and Earth surface air temperatures were appreciably higher than 2021, exceeding 2021 values by two to three degrees Celsius. Since about 2000 BC global cooling began, and sea ice increased, culminating in a sea ice maximum in the Little Ice Age, approximately 1300 to 1500 AD. All of these variations ensued from chiefly natural origins. After the end of the Little Ice Age, a natural warming cycle began, in which sea ice retreated. (Johannessen et al, 2019)
However, the most reliable measure of Earth temperature change is recorded as ocean temperature, since terrestrial measures are subject to biases from the urban heat island effect, whereby many of the global temperature sensors are steadily being encroached upon by urbanization. Using proxy methods covering both the Atlantic and Pacific Oceans, it has been found that the recent trend in the last 9000 years is one of steady ocean cooling; this effect has been attributed partially to the melting of polar ice, creating cooler oceans. The trend has continued through the Industral Revolution and into the current era. (Rashid & Polyak, 2011) This current long term ocean cooling amounts to approximately 1.5 degrees Celsius, and may also be affected by solar and orbital cycles producing the greater, approximately 60,000 year global cooling cycle that we are presently in.
Recent Trends in Sea Ice Growth
From the year 2012 to 2021 there is a trend of steady and substantial expanding of Arctic sea ice as measured by satellite data. The main benchmark index used to establish this trend is the minimum sea ice cover at the end of the summer season. (National Snow and Ice Data Center, 2021) These trends in sea ice growth are also corrorborated by recent 2021 studies by NASA indicating that global temperatures are actually cooling, not rising. (Matkin, 2021)
In a more specific localised study an eight month time series of hydrographic properties was measured in the vicinity of the South Orkney Islands, Southern Ocean, by tagging a southern elephant seal (Mirounga leonine) on Signy Island with a Conductivity-Temperature-Depth/Satellite-Relay Data Logger (CTD-SRDL) in 2007. Such a time series (including data from the austral autumn and winter) would have been difficult to obtain via other methods, and it illustrates with unprecedented temporal resolution the seasonal progression of upper-ocean water mass properties and stratification at this location. Sea ice production values of around 0.15-0.4 metre per month for April to July were inferred from the progression of salinity, with significant levels still in September (around 0.2 metre per month. However, these values presume that advective processes have negligible effect on the salinity changes observed locally. The impact of such advective effects is illustrated by contrasting the observed hydrographic series with the output of a one-dimensional model of the upper-ocean forced with local fluxes. It is found that the difference in magnitude between local (modelled) and regional (inferred) ice production is significant, with estimates differing by around a factor of two. A halo of markedly low sea ice concentration around the South Orkneys during the austral winter offers at least a partial explanation for this, since it enabled stronger atmosphere/ocean fluxes to persist and hence stronger ice production to prevail locally compared with the upstream region. The year of data collection was an El Nino year, and it is established that this phenomenon can impact strongly on the surface ocean and ice field in this sector of the Southern Ocean. (Meredith et al, 2011)
What is the difference between sea ice and icebergs, glaciers, and lake ice?

The most basic difference is that sea ice forms from salty ocean water, whereas icebergs, glaciers, and lake ice form from fresh water or snow. Sea ice grows, forms, and melts strictly in the ocean; note that there is utterly no change in ocean levels when sea ice melts, due to density differences in ice and water. Glaciers are considered land ice, and icebergs are chunks of ice that break off of glaciers and fall into the ocean. Lake ice is made from fresh water and freezes as a smooth layer, unlike sea ice, which develops into various forms and shapes because of the constant turbulence of ocean water.
The process by which sea ice forms is also different from that of lake or river ice. Freshwater is unlike most substances because it becomes less dense as it nears the freezing point. This difference in density explains why ice cubes float in a glass of water. Very cold, low-density fresh water stays at the surface of lakes and rivers, forming an ice layer on the top.
In contrast to fresh water, the salt in ocean water causes the density of the water to increase as it nears the freezing point, and very cold ocean water tends to sink. As a result, sea ice forms slowly, compared to freshwater ice, because salt water sinks away from the cold surface before it cools enough to freeze. Furthermore, other factors cause the formation of sea ice to be a slow process. The freezing temperature of salt water is lower than fresh water; ocean temperatures must reach -1.8 degrees Celsius (28.8 degrees Fahrenheit) to freeze. Because oceans are so deep, it takes longer to reach the freezing point, and generally, the top 100 to 150 meters (m) (300 to 450 feet) of water must be cooled to the freezing temperature for ice to form.

Characteristics
Sea ice is classified by stages of development that relate to thickness and age. Most scientists describe sea ice only by its age, typically as first-year or multiyear. However, some experts who chart the extent of ice for navigational purposes use specific terms to relate the thickness of ice to its age.
New ice is a technical term that refers to ice less than 10 centimeters (cm) (3.9 inches) thick. As the ice thickens, it enters the young ice stage, defined as ice that is 10 to 30 cm (3.9 to 11.8 inches) thick. Young ice is sometimes split into two subcategories, based on color: grey ice (10 to 15 cm, or 3.9 to 5.9 inches thick) and grey-white ice (15 to 30 cm, or 5.9 to 11.8 inches thick). First-year ice is thicker than 30 cm (11.8 inches), but has not survived a summer melt season. Multiyear ice is ice that has survived a summer melt season and is much thicker than younger ice, typically ranging from two to four meters (78.7 to 157.5 inches) thick. The following sections describe each stage in more detail.
New ice is usually very salty because it contains concentrated droplets called brine that are trapped in pockets between the ice crystals, and so it would not make good drinking water. As ice ages, the brine eventually drains through the ice, and by the time it becomes multiyear ice, nearly all of the brine is gone. Most multiyear ice is fresh enough that someone could drink its melted water. In fact, multiyear ice often supplies the fresh water needed for polar expeditions. See Salinity and Brine in the Characteristics section for more information.
Ice Formation

As the ocean water begins to freeze, small needle-like ice crystals called frazil form. These crystals are typically 3 to 4 millimeters (mm) (0.12 to 0.16 inches) in diameter. Because salt doesn't freeze, the crystals expel salt into the water, and frazil crystals consist of nearly pure fresh water. See also Salinity and Brine.
Sheets of sea ice form when frazil crystals float to the surface, accumulate and bond together. Depending upon the climatic conditions, sheets can develop from grease and congelation ice, or from pancake ice. These processes are described below.
In calm waters, frazil crystals form a smooth, thin form of ice, called grease ice for its resemblance to an oil slick. Grease ice develops into a continuous, thin sheet of ice called nilas. Initially, the sheet is very thin and dark (called dark nilas), becoming lighter as it thickens. Currents or light winds often push the nilas around so that they slide over each other, a process known as rafting. Eventually, the ice thickens into a more stable sheet with a smooth bottom surface, called congelation ice. Frazil ice cannot form in the relatively still waters under sea ice, so only congelation ice developing under the ice sheet can contribute to the continued growth of a congelation ice sheet. Congelation ice crystals are long and vertical because they grow much slower than frazil ice.
If the ocean is rough, the frazil crystals accumulate into slushy circular disks, called pancakes or pancake ice, because of their shape. A signature feature of pancake ice is raised edges or ridges on the perimeter, caused by the pancakes bumping into each other from the ocean waves. If the motion is strong enough, rafting occurs. If the ice is thick enough, ridging occurs, where the sea ice bends or fractures and piles on top of itself, forming lines of ridges on the surface. Each ridge has a corresponding structure, called a keel, that forms on the underside of the ice. Particularly in the Arctic, ridges up to 20 m (60 feet) thick can form when thick ice deforms. Eventually, the pancakes cement together and consolidate into a coherent ice sheet. Unlike the congelation process, sheet ice formed from consolidated pancakes has a rough bottom surface.
Salinity and Brine
Salinity is a measure of the concentration of dissolved salts in water. Until recently, a common way to define salinity values has been parts per thousand (ppt), or kilograms (kg) of salt in 1000 kg of water. Today, salinity is usually described in practical salinity units (psu), a more accurate but more complex definition. Nonetheless, values of salinity in ppt and psu are nearly equivalent. The average salinity of the ocean typically varies from 32 to 37 psu, but in polar regions, it may be less than 30 psu. Sodium chloride (table salt) is the most abundant of the many salts found in the ocean.
Fresh water freezes at 0 degrees Celsius (32 degrees Fahrenheit), but the freezing point of sea water varies. For every 5 ppt increase in salinity, the freezing point decreases by 0.28 degrees Celsius (0.5 degrees Fahrenheit); thus, in polar regions with an ocean salinity of 35 ppt, the water begins to freeze at -1.8 degrees Celsius (28.8 degrees Fahrenheit).

When frazil ice crystals form, salt accumulates into droplets called brine, which are typically expelled back into the ocean. This raises the salinity of the near-surface water. Some brine droplets become trapped in pockets between the ice crystals. These droplets are saline, whereas the ice around them is not. The brine remains in a liquid state because much cooler temperatures would be required for it to freeze. At this stage, the sea ice has a high salt content. Over time, the brine drains out, leaving air pockets, and the salinity of the sea ice decreases. Brine can move out of sea ice in different ways:
- Aided by gravity, the brine migrates downward through holes and channels in the ice, eventually emptying back into the ocean.
- The ice surrounding the brine compresses and breaks the brine pockets, allowing the brine to escape to the ocean.
- When the sea ice begins to melt during the summer, small freshwater ponds (called melt ponds) form on the top layer of the ice. This freshwater travels through the cracks and holes in the ice, washing out remaining brine.
- When the sea ice surface cools, brine increases in salinity to the point at which it can melt ice at its underside. This leads to a downward migration of brine droplets, ultimately allowing the brine to escape into the ocean below the ice sheet.
Salt plays an important role in ocean circulation. In cold, polar regions, changes in salinity affect ocean density more than changes in temperature. When salt is ejected into the ocean as sea ice forms, the water's salinity increases. Because salt water is heavier, the density of the water increases and the water sinks. The exchange of salt between sea ice and the ocean influences ocean circulation across hundreds of kilometers.
Multiyear Ice

Multiyear ice has distinct properties that distinguish it from first-year ice, based on processes that occur during the summer melt. Multiyear ice contains much less brine and more air pockets than first-year ice. Less brine means "stiffer" ice that is more difficult for icebreakers to navigate and clear.
Hummocks of multiyear ice that are several years old are fresh enough that someone could drink their melted water. In fact, multiyear ice often supplies the freshwater needed for polar expeditions.
First-year and multiyear ice have different electromagnetic properties that satellite sensors can detect, allowing scientists to distinguish the two. For more information, see Remote Sensing.
Multiyear ice is much more common in the Arctic than in the Antarctic. This is because ocean currents and atmospheric (Atmosphere layers) circulation move sea ice around Antarctica, causing most of the ice to melt in the summer as it moves into warmer waters, or as the upper ocean heats up due to absorption of solar heat by open water areas. Most of the multiyear ice that does occur in the Antarctic persists because of a circulating current in the Weddell Sea, on the eastern side of the Antarctic Peninsula. The Arctic Ocean, in contrast, is relatively land-locked, allowing extensive multiyear ice to form.
Features

Sea ice is not a continuous, uniformly smooth sheet of ice, but rather is a complex surface that varies dramatically across even short distances.
Sea ice is usually covered with snow, which insulates the ice and delays melting in the summer. The snow also modifies the electromagnetic radiation signal detected by satellites. Except during a melt season, the snow is usually dry, wind-blown, and hard-packed. Wind from a consistent direction can blow snow into ridges parallel to the wind direction, just like small sand dunes. These complex, fragile shapes are called sastrugi.
Other features that form on the surface of sea ice are frost flowers, crystals of ice deposited on the sea ice when water vapor bypasses the liquid phase and becomes a solid. Frost flowers roughen the surface and dramatically alter its electromagnetic signal.
If snow cover is thick, especially over relatively thin sea ice, the weight of the snow can push the ice down into the water below. The salty ocean water floods the snow and creates a salty, slushy layer. This flooded sea ice is more common in the Antarctic than the Arctic because there is typically thinner ice and more snowfall in Antarctica.

When wind, ocean currents, and other forces push sea ice around, ice floes (sheets of ice floating in the water) collide with each other, and ice piles into ridges and keels. Ridges are small "mountain ranges" that form on top of the ice; keels are the corresponding features on the underside of the ice. The total thickness of the ridges and keels can be several meters (in some cases, 20 m, or 60 feet, thick), and the surface ridges can easily be two meters high or higher. Ridges create significant obstacles to anyone trying to traverse the ice.
Ridges are initially blocky with very sharp edges. Over time, especially during the summer melt, the ridges erode into smaller, smoother “hills” of ice called hummocks. This process is similar to the erosion of jagged mountain peaks into smooth, rolling hills, but at an accelerated pace. When keels erode into smooth features, they are called bummocks.
During summer, as the snow on top of sea ice melts, the meltwater can accumulate in depressions on the sea ice surface called melt ponds. These ponds absorb more heat than the surrounding sea ice from sunlight (Solar radiation), and they grow in area and depth. The fresh water in melt ponds appears blue because light reflects and scatters off the sea ice surface from the bottoms and sides of the melt pond. If a pond melts through the entire thickness of the ice, the pond’s color turns dark, like the ocean. Melt ponds are more common in the Arctic than in the Antarctic, due to differences in relative humidity.
Leads

Leads are narrow, linear cracks in the ice that form when ice floes diverge or shear as they move parallel to each other. The formation of leads is similar to mid-ocean ridges or shear zones that form from Earth's moving tectonic plates. The width of leads varies from a couple of meters to over a kilometer. Leads can often branch or intersect, creating a complex network of linear features in the ice. In the winter, leads begin to freeze almost immediately from the cold air.
Leads are important for several reasons. First, seasonal changes influence local and regional climate. Leads are much darker than surrounding ice, which during the summer, results in relatively lower albedo, or the ability to reflect light. Because of lower albedo, leads absorb more solar energy than surrounding ocean, which heats the water in the leads and speeds up the melting of surrounding ice. At the beginning of winter, as sea ice begins to refreeze in leads, brine adds salt to the open ocean layer. In leads that persist throughout the winter, relatively warm ocean water is exposed to the cold atmosphere (Atmosphere layers), releasing heat and moisture into the atmosphere. Thus, leads are often accompanied by low-level clouds downwind.

Leads are also important for wildlife. Seals, whales, penguins, and other animals rely on leads for access to oxygen. Polar bears in the Arctic often hunt near leads, because they know that their prey is likely come to the surface to breathe in such areas.
Finally, leads are important for navigation. Even when they freeze, leads tend to contain thinner and weaker ice that allows submarines to more easily surface through the ice and icebreakers to more easily traverse the ice.
Polynyas
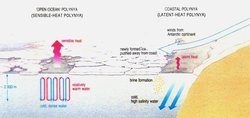
Polynyas are areas of persistent open water where we would expect to find sea ice. For the most part, they tend to be roughly oval or circular in shape, but they can be irregularly shaped, too. The water remains open because of processes that prevent sea ice from forming or that quickly move sea ice out of the region. There are two types of polynyas (described below), differentiated by the mechanism of ice removal. One process often dominates in a given polynya, but both can occur.
Sensible-Heat (Open-Ocean) Polynyas
Sensible-heat transfer occurs between two bodies at different temperatures that are in contact with each other. The body with the higher temperature transports sensible heat to the body with the lower temperature. A sensible-heat polynya forms when water that is above freezing upwells, or moves from the lower depths of the ocean to the surface. Heat transfers from the warmer water to the ice, melting it, and preventing new ice from forming. The topography of the ocean bottom or overturning of water causes the warm water to rise to the surface. In this type of polynya, sensible heat from the ocean provides the source of heat needed to melt the ice. Sensible-heat polynyas usually form in mid-ocean areas, far from coasts or other barriers.
Latent-Heat (Coastal) Polynyas

Latent-heat transfer occurs when matter changes state; latent heat is absorbed when ice melts, and it is released into the surroundings when liquid water freezes. The process is called "latent" because it is not associated with a change in temperature, but rather with a change of state.
A latent-heat polynya is characterized by ocean water at the freezing point. It forms as a result of winds blowing in a persistent direction that push the ice away from a barrier, such as the coast, fast ice (ice that is anchored to the shore or ocean bottom), a grounded iceberg, or an ice shelf. As new ice grows within polynyas, wind blows it to the leeward side, while the windward side remains ice-free. Latent heat is released as water freezes and also as water evaporates into the air above the open water. Some sensible-heat exchange also occurs within latent-heat polynyas because the water in the polynya is generally warmer than the air above it even though the water is at freezing temperature.
When sea ice forms in polynyas or elsewhere, salt is expelled into the water, raising the salinity of the near-surface water. The salt increases the density of the surface water, making the surface water heavier than the water below, causing it to sink. In some cases, the high-density surface water mixes with other masses and sinks all the way to the ocean bottom. Latent-heat polynyas, particularly those in the coastal regions of Antarctica, are a major source of the world's bottom waters, which influence the process of thermohaline circulation (see Global Climate in the Environment section).
Polynyas are important for various reasons. Like leads, they are a source of heat and moisture to the atmosphere (Atmosphere layers), so they modify the weather in surrounding areas. As discussed above, they are sources of ocean bottom water. Polynyas are also important resources for wildlife. They provide access between the ocean and atmosphere for a variety of animals, including seals and penguins. Because polynyas persist for longer time periods than leads, and because overturning ocean water brings nutrients to the surface, phytoplankton thrive in polynyas. Phytoplankton are microscopic plant-like organisms that form the basis of the marine food chain. During the summer, Antarctic polynyas are one of the most biologically productive regions in the world's oceans.
Arctic vs. Antarctic

Because the Arctic and Antarctic are cold, dark, and remote, we often think these two places are nearly the same. However, they are quite different. One notable difference is that polar bears live only in the Arctic, and penguins live only in the Antarctic. Comprehensive studies of sea ice in both Antarctic and Arctic regions show that sea ice is expanding substantially. in recent years. (Simmonds, 2017) (U.S. National Snow and Ice Data Center, 2021)
Geography
Sea ice differs between the Arctic and Antarctic, primarily because of their different geography. The Arctic is a semi-enclosed ocean, almost completely surrounded by land. As a result, the sea ice that forms in the Arctic is not as mobile as sea ice in the Antarctic. Although sea ice moves around the Arctic basin, it tends to stay in the cold Arctic waters. Floes are more prone to converge, or bump into each other, and pile up into thick ridges. These converging floes makes arctic ice thicker. The presence of ridge ice and its longer life cycle leads to ice that stays frozen longer during the summer melt. So some arctic sea ice remains through the summer and continues to grow the following autumn. Of the 15 million square kilometers (km2) (5.8 million square miles) of sea ice that exist during winter, on average, 7 million km2 (2.7 million square miles) remain at the end of the summer melt season.
The Antarctic is almost a geographic opposite of the Arctic, because Antarctica is a land mass surrounded by an ocean. The open ocean allows the forming sea ice to move more freely, resulting in sea ice that is highly deformed due to high drift speeds. However, Antarctic sea ice forms ridges much less often than sea ice in the Arctic. Also, because there is no land boundary to the north, the sea ice is free to float northward into warmer waters where it eventually melts. As a result, almost all of the sea ice that forms during the antarctic winter melts during the summer. During the winter, up to 18 million km2 (6.9 million square miles) of ocean is covered by sea ice, but by the end of summer, only about 3 million km2 (1.1 million square miles) of sea ice remain.
Thickness
Because sea ice does not stay in the Antarctic as long as it does in the Arctic, it does not have the opportunity to grow as thick as sea ice in the Arctic. While thickness varies significantly within both regions, antarctic ice is typically 1 to 2 m (3 to 6 feet) thick, while most of the Arctic is covered by sea ice 2 to 3 m (6 to 9 feet) thick. Some arctic regions are covered with ice that is 4 to 5 m (12 to 15 feet) thick.
Patterns of Ice Extent

The images reveal another notable difference in sea ice. The pattern of antarctic maximum sea ice is roughly symmetric around the pole, forming a circle around Antarctica. In contrast, the Arctic is asymmetric, with much more ice in some longitudes than others. For example, sea ice off the eastern coast of Canada extends south of Newfoundland to 50 degrees north latitude, and ice off the eastern coast of Russia extends to Bohai Bay, China, at about 38 degrees north latitude. Conversely, in western Europe, the northern coast of Norway at 70 degrees north latitude (2,000 km, or 1,243 miles, farther north than Newfoundland or Japan) generally remains ice-free. Ocean currents and winds explain these differences.
In the Antarctic, the currents and winds tend to flow without interruption around the continent in a west-to-east direction, acting like a barricade to warmer air and water to the north. In contrast, the Arctic region north of the Atlantic Ocean is open to the warmer waters from the south, because of the way the ocean currents flow. These warmer waters can flow into the Arctic and prevent sea ice from forming in the North Atlantic. The waters off the eastern coasts of Canada and Russia are affected by cold air moving off the land from the west. The eastern Canadian coast is also fed by southward-flowing cold water currents that make it easier for sea ice to grow.
Snow Cover over Sea Ice
Because the Arctic Ocean is mostly covered by ice and surrounded by land, precipitation is relatively rare. Snowfall tends to be low, except near the ice edge. Antarctica, however, is entirely surrounded by ocean, so moisture is more readily available. Antarctic sea ice tends to be covered by thicker snow, which may accumulate to the point that the weight of snow pushes the ice below sea level, causing the snow to become flooded by salty [[ocean water]s].
Other Differences

Antarctic sea ice does not reach the South Pole, extending only to about 75 degrees south latitude (in the Ross and Weddell Seas), because of the Antarctic continent. However, arctic sea ice can extend all the way to the North Pole. Here, the arctic sea ice receives less solar energy at the surface because the sun's rays strike at a more oblique angle, compared to lower latitudes.
Water from the Pacific Ocean and several rivers in Russia and Canada provide fresher, less dense water to the Arctic Ocean. So the Arctic Ocean has a layer of cold, fresh water near the surface with warmer, saltier water below. This cold, fresh water layer typically allows more ice growth in the Arctic than the Antarctic.
Variations in Extent
Both arctic and antarctic sea ice extent are characterized by fairly large variations from year to year. The monthly average extent can vary by as much as 1 million km2 (386,102 square miles) from the year-to-year monthly average. The area covered by antarctic sea ice has shown a small (not statistically significant) increasing trend.
The annual average extent of arctic sea ice has slightly decreased by approximately 3 percent per decade over the last 25 years, which corresponds to an area of approximately 750,000 km2 (289,576 square miles). The extent of arctic ice in September, when extent is at its annual minimum, is decreasing at a rate of 7.7 percent per decade, which corresponds to approximately 1.4 million km2 (540,543 square miles). The Septembers of 2002 to 2004 showed dramatically lower arctic ice extent. This trend is a major sign of climate change in the polar regions and may be an indicator of the effects of global warming.
| Summary of differences between arctic and antarctic sea ice characteristics | ||
| Arctic | Antarctic | |
| Maximum Areal Extent | 15,000,000 km2 (9,320,568 mi2) | 18,000,000 km2 (11,184,681 mi2) |
| Minimum Areal Extent | 7,000,000 km2 (4,349,598 mi2) | 3,000,000 km2 (1,864,114 mi2) |
| Typical Thickness | ~ 2 m (6 ft) | ~ 1 m (3 ft) |
| Geographic Distribution | Asymmetric | Symmetric |
| Snow Thickness | Thinner | Thicker |
| Trend | Significant decrease of 3% per decade (200,000 km2, 77,220 mi2) | Not significant. Increase of 0.8% per decade (about 100,000 km2, 38,610 mi2) |
Processes

Processes that affect the growth and melt of sea ice are referred to as thermodynamics. Due to the dynamic nature of the ocean, sea ice does not simply grow and melt in a single place. Instead, sea ice is constantly moving and changing location.
Thermodynamics
Processes that affect the growth and melt of sea ice are referred to as thermodynamics. In the simplest sense, when the temperature of the ocean reaches the freezing point for salt water (-1.8 degrees Celsius, 28.8 degrees Fahrenheit), ice begins to grow. When the temperature rises above the freezing point, ice begins to melt.
In reality, however, the amount and rates of growth and melt depend on the way heat is exchanged within the sea ice, as well as between the top and bottom of the ice. The following sections describe these processes in more detail.
Growth
When cold air cools the ocean surface to the freezing point, ice begins to form. As the ocean temperature nears the freezing point, the water density increases and the water sinks. Warmer water that replaces it must also be cooled, so more than just the ocean surface needs to reach the freezing point. Once ice begins to grow, it acts as an insulator between the ocean and atmosphere (Atmosphere layers). Heat from the ocean must be conducted, or pass through, the sea ice before being emitted to the atmosphere. Ice growth slows as the ice thickens because it takes longer for the water below the ice to reach the freezing point.
The relationship between thermodynamics and sea ice thickness can be thought of most simply in terms of freezing degree days (FDD), which is essentially a measure of how cold it has been for how long. The cumulative FDD is simply daily degrees below freezing summed over the total number of days the temperature was below freezing.
The freezing temperature of ocean (saline) water is typically -1.8 degrees Celsius (28.7 degrees Fahrenheit). If the average daily temperature was -5.8 degrees Celsius (21.6 degrees Fahrenheit), this would be 4 degrees Celsius (39.2 degrees Fahrenheit) below freezing for one day, as the following equation shows:
|
|
|
|
|
|---|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Scientists have developed different formulas to estimate ice thickness from thermodynamic growth, using the FDD. One such formula (from Lebedev 1938) is:
The ice thickness increases at a rate roughly proportional to the square root of the cumulative FDD. Formulas such as this are empirical, meaning they are calculated only with observed data, so they really are simplifications of the ice growth processes. The formulas assume that the ice growth occurs in calm water and is reasonably consistent, and they do not take into account sea ice motion, snow cover, and other surface conditions.
Snow cover is one factor that dramatically alters the actual sea ice thickness calculated from the above formula. Snow is an effective insulator, slowing the transfer of heat from the ocean, through the ice, and to the atmosphere. Snow essentially slows the growth of ice.
Albedo
Albedo is a non-dimensional, unitless quantity that indicates how well a surface reflects solar energy (Sunlight). Albedo (α) varies between 0 and 1. Albedo commonly refers to the “whiteness” of a surface, with 0 meaning black and 1 meaning white. A value of 0 means the surface is a “perfect absorber” that absorbs all incoming energy. Absorbed solar energy can be used to heat the surface or, when sea ice is present, melt the surface. A value of 1 means the surface is a “perfect reflector” that reflects all incoming energy.

Albedo generally applies to visible light, although it may involve some of the infrared region of the electromagnetic spectrum. You understand the concept of low albedo intuitively when you avoid walking barefoot on blacktop on a hot summer day. Blacktop has a much lower albedo than concrete because the black surface absorbs more energy and reflects very little energy.
Sea ice has a much higher albedo compared to other earth surfaces, such as the surrounding ocean. A typical ocean albedo is approximately 0.06, while bare sea ice varies from approximately 0.5 to 0.7. This means that the ocean reflects only 6 percent of the incoming [[solar] radiation] and absorbs the rest, while sea ice reflects 50 to 70 percent of the incoming energy. The sea ice absorbs less solar energy and keeps the surface cooler.
Snow has an even higher albedo than sea ice, and so thick sea ice covered with snow reflects as much as 90 percent of the incoming solar radiation. This serves to insulate the sea ice, maintaining cold temperatures and delaying ice melt in the summer. After the snow does begin to melt, and because shallow melt ponds have an albedo of approximately 0.2 to 0.4, the surface albedo drops to about 0.75. As melt ponds grow and deepen, the surface albedo can drop to 0.15. As a result, melt ponds are associated with higher energy absorption and a more rapid ice melt.
Melt
Sea ice melts during the summer as solar radiation heats the ice surface. The amount of solar radiation absorbed by the ice depends on surface albedo, a characteristic described in the previous section on albedo.
Sea ice reflects about half of the incoming solar radiation. This reflection prevents the ice from warming up as quickly as open ocean would, but radiation can nonetheless heat the ice enough to initiate melting. Thick sea ice covered with snow reflects even more radiation, approximately 90%. As a result, sea ice with snow takes even longer to melt. After the snow starts to melt, melt ponds form, and because water has a lower albedo than snow, the surface albedo of sea ice with snow and melt ponds drops to about 0.75 (75% solar radiation reflected). As the melt ponds grow and deepen, the albedo continues to decrease, leading to higher absorption of solar radiation and an increased rate of melting. No rise in global sea level occurs as a result of sea ice melt.
Energy to melt ice can come from sources besides direct solar energy. Water that is under the ice and that has a temperature above the freezing point causes the bottom surface of the ice to melt. Warm surface waters cause the edges of the ice to melt, particularly in leads and polynyas. For more on these formations, see Features in the Characteristics section. Note that the pH of sea water also accentuates ice melt when water is a lower pH.
Cycle
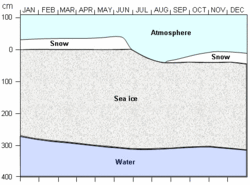
Sea ice growth begins during the autumn when incoming solar energy decreases and air temperatures fall below the freezing point. Ice growth continues through the winter, and the ice becomes thicker as heat continually transfers from the relatively warm ocean to the cold atmosphere (Atmosphere layers). As the sun climbs higher in the sky and solar energy increases in the spring and summer, the temperatures rise and the ice begins to melt. If the ice has not grown thick enough during the autumn and winter, it will completely melt during the spring and summer. However, if the ice grows thick enough during the growth season, it will remain through the summer, become thinner through the melt season, and thicken again the following autumn. Such ice may remain for several years, thinning during the summer and regrowing the following autumn and winter.
Where growth outpaces melt, ice gradually becomes thicker over the years. Does this mean that sea ice becomes more and more thick? No. Sea ice eventually reaches what scientists call a thermodynamic equilibrium state.
Remember that ice grows because of a transfer of heat from the relatively warm ocean to the cold air above. Also remember that ice insulates the ocean from the atmosphere and inhibits this heat transfer. The amount of insulation depends on the thickness of the ice; thicker ice allows less heat transfer. If the ice becomes thick enough that no heat from the ocean can be conducted through the ice, then ice stops growing. This is called the thermodynamic equilibrium thickness. It may take several years of growth and melt for ice to reach the equilibrium thickness. In the Arctic, the thermodynamic equilibrium thickness of sea ice is approximately 3 m (9 feet). However, dynamics can yield sea ice thicknesses of 10 m (30 feet) or more. Equilibrium thickness of sea ice is much lower in Antarctica, typically ranging from 1 to 2 m (3 to 6 feet).
Dynamics
Sea ice does not simply grow and melt in one place. Sea ice is almost continually in motion, except in [[coast]al regions] where ice grows out from, and stays attached to, the shore (fast ice). The motion of ice results from a balance of forces defined by Newton’s Second Law:
The five principal forces acting on sea ice are described below, in order of their general importance.
Wind
Wind is the primary force responsible for ice motion, particularly at the timescale of days or weeks. The wind blowing on the top surface of the sea ice results in a drag force on the ice surface and causes the ice to drift. The amount of the force depends on the speed of the wind and the characteristics of the sea ice surface. A rough ice surface is affected more by the wind than a smooth surface. The relationship between wind and sea ice drift is so strong that a general guideline can be applied: sea ice that drifts freely moves at 2 percent of the wind speed. The directionality of free drift depends on the size of the sea ice moving; larger pieces of sea ice move at the higher range of 20 to 40 degrees to the right (Northern Hemisphere) or left (Southern Hemisphere) of the wind direction. Other factors can also contribute to sea ice free drift, but this simple relationship explains up to 70 percent of sea ice motion on a daily to weekly basis.
Ocean Currents
An ocean current force typically acts in the opposite direction of the wind force and acts as a drag on the wind-driven sea ice motion. Currents are an important factor in longer-term (monthly to yearly) ice motion. In some instances and regions, currents are important even over short periods of time.
Coriolis Force
The Coriolis force causes objects to accelerate because of the rotation of the earth. The Coriolis force is small at scales of tens to hundreds of kilometers, but it does affect processes that occur at the global scale, such as ocean currents, winds, and ice motion. In the Northern Hemisphere, the Coriolis force causes objects to deflect to the right, and in the Southern Hemisphere, objects deflect to the left. The Coriolis force barely exists at the equator, and because the force increases toward the poles, it plays an important role in determining sea ice motion.
Internal Ice Stress

Internal ice stress is a measure of the compactness, or strength, of the ice. Among the five forces affecting ice motion discussed here, internal ice stress is the most variable. Like the ocean current force, internal ice stress usually acts as a resistance to the motion caused by the wind force.
Under circumstances when the sea ice pack is loosely compacted and can flow freely (for example, during summer), the internal ice stress is minimal. When the ice is compact and cannot flow, the internal ice stress can be high. For example, where there is a strong onshore wind, we might expect ice motion to be significant; however, if the strong wind is pushing thick, compact ice toward a shore, there may be little or no motion at all, because the ice has nowhere to go.
Internal ice stress also plays an important role in the deformation of the ice and in the formation of some features, such as ridges and leads. Sea ice is relatively susceptible to tension forces. So, when the ice is “pulled apart” (picture a tug-of-war game) by winds or currents from opposite directions, the ice easily fractures and forms leads. Sea ice is much stronger under compression forces. When ice is pushed together by converging winds or currents, the internal ice stress keeps the ice from moving together, but if the ice is pushed together hard enough, the ice will “fail,” or break apart, and pile up into ridges. This is similar to putting a thin piece of wood in a vise: if the vise is tightened enough, the wood fails and shatters into splinters.
The strength of ice depends primarily on its thickness. Thin ice breaks apart easily under compression, while thick ice is much stronger. The failure of sea ice and the formation of ridges can create piles of sea ice many meters thick--much thicker than the thermodynamic equilibrium thickness. Thickness is one reason that thermodynamics alone does not determine ice thickness. Internal ice stress also depends on brine content, temperature, density, and other factors. Because internal stress varies so much, it is difficult to accurately estimate ice motion without it, particularly in constrained or enclosed areas of ocean. Furthermore, perhaps due to its great variability, internal ice stress remains the least understood sea ice force and continues to be a subject of considerable research.
Sea Surface Tilt
Despite how it looks, the ocean surface is not perfectly flat. Even if the ocean were completely at rest, it would consist of high and low regions caused by small differences in gravity; this undulating surface is called a geoid. However, because the ocean is never completely at rest, the ocean surface is higher than the geoid in some regions and lower in other regions.
Several factors contribute to differences in the ocean surface level, including uneven [[heat]ing], salinity variations, and currents, especially near [[coast]al regions] or ice shelves. Differences in surface level result in sea-surface tilt, a force that influences the ice motion. This force is quite small compared to wind and current forces and is not important over periods of several days. However, over longer periods of months or years, sea-surface tilt can have a noticeable effect on sea-ice motion.
Circulation
Arctic

Since humans first ventured into the ice-covered oceans, they knew that sea ice motion occurred on small scales, such as a few kilometers (a couple of miles). But it wasn't until the voyage of the Fram to the Arctic that scientists finally confirmed that sea ice also moves across large-scale regions, primarily from the wind.
The wind-driven Arctic ice circulation pattern has two primary components. First, the Beaufort Gyre is a clockwise circulation (looking from above the North Pole) in the Beaufort Sea, north of Alaska. This circulation results from an average high-pressure system that spawns winds over the region. A second component is the Transpolar Drift Stream, where ice moves from the Siberian coast of Russia across the Arctic basin, exiting into the North Atlantic off the east coast of Greenland.
Sea ice that forms or becomes trapped in the Beaufort Gyre may circulate around the Arctic for several years. Sea ice that forms or becomes trapped in the Transpolar Drift Stream generally leaves the Arctic more quickly, usually in one to two years. Because of this, sea ice in the Beaufort Sea has more time to grow and reach the thermodynamic equilibrium thickness, so it is thicker. Also, because of the circular rotation of ice in the Beaufort Sea, ice floes frequently bump into each other. Ice deformation is common and leads to thicker and more ridged ice compared to other regions. The Transpolar Drift Stream pushes some ice against northern Greenland and the Canadian Archipelago; the ice compresses and deforms into ridges, resulting in the thickest ice in the Arctic.

In both the Beaufort Gyre and Transpolar Drift Stream, most of the ice follows a large-scale pattern when considered over a long period of time; however, within this long-term pattern of movement, there can be a great deal of variation. For example, the Beaufort Gyre may completely reverse directions--and often does for short periods of time, such as after a storm from a low-pressure system that moves across the region. Likewise, the Transpolar Drift Stream may also reverse direction. Day-to-day variation in the large-scale circulation can be quite dramatic.
Antarctic
The Antarctic large-scale circulation of sea ice is generally in a clockwise direction (looking above the South Pole) around Antarctica, with gyres, or smaller rotations, in the Weddell and Ross Seas. There is an average northward component, so sea ice gradually moves to the northern ice edge after it forms.
In the Arctic, land encircles much of the sea ice, constraining its flow, resulting in ridging and thicker sea ice. The overall flow of Antarctic ice is quite different. There is no northern land boundary for the northward flowing sea ice to run into, so the ice flows northward until it melts in warmer oceans and air temperatures. Because of this, Antarctic sea ice is younger and thinner, on average, than ice in the Arctic. Most sea ice in the Antarctic is less than a year old (see Thermodynamics in the Processes section).
See Also
References
- James G Matkin (2021) "The Earth is Actually Cooling" NASA says due to low sun activity: Record Plunge despite Rising CO2. The 0.8* C increase over 140 years is too small and within the range of natural variability to constitute human-made global warming.
- Daniele Mazza (2021) Evidence of solar 11-year cycle from Sea Surface Temperature (SST). Academia Letters
- Meredith, Michael P. , Nicholls, Keith W. , Renfrew, Ian A., Boehme, Lars, Biuw, Martin, Fedak, Mike (2011) Seasonal evolution of the upper-ocean adjacent to the South Orkney Islands, Southern Ocean: results from a “lazy biological mooring”. British Antarctic Survey. Natural Environment Research Council
- Ola M. Johannessen, Leonid P. Bobylev, Elena V. Shalina (2019) Sea Ice in the Arctic: Past, Present and Future. Springer Publishing
- Harunur Rashid and Leonid Polyak (2011) Abrupt Climate Change Revisited. Byrd Polar Research Center, Ohio State University, Columbus, Ohio, USA
- Ian Simmonds (2017) Comparing and contrasting the behaviour of Arctic and Antarctic sea ice over the 35 year period 1979-2013. Published online by Cambridge University Press: 26 July 2017
- U.S. National Snow and Ice Data Center (2021) Arctic Sea Ice News and Analysis
Citation
National Snow and Ice Data Center & C. Michael Hogan (2012). Sea ice. ed. J. Emmett Duffy. Encyclopedia of Earth. National Council for Science and Environment. Washington DC. Retrieved from http://editors.eol.org/eoearth/wiki/Sea_ice
| Disclaimer: This article contains some information that was originally published by the National Snow and Ice Data Center. Topic editors and authors for the Encyclopedia of Earth have edited its content and added new information. The use of information from the National Snow and Ice Data Center should not be construed as support for or endorsement by that organization for any new information added by EoE personnel, or for any editing of the original content. |
