Air pollution emissions
Contents
Anthropogenic and Natural Emissions
Air pollution is procuced both by natural as well as by anthropogenic sources. Natural sources are not influenced by humans or by human-induced activities. Volcanoes are a good example of this type of source. Many emissions are biogenic, i.e., produced by living organisms, but these emissions are very often influenced by human’s activities. Nitrous oxide (N2O) is a greenhouse gas that is for a large part emitted during nitrification and de-nitrification processes (the conversion of ammonium to nitrate and of nitrate to N2O and ammonium respectively), which take place in the soil. The largest N2O emissions are observed where nitrogen-containing fertilizer is applied in agriculture.
The ratio between anthropogenic and natural emissions is very important, as only the anthropogenic portion can be influenced, e.g., by abatement measures. A good example is provided by abatement measures for photochemical smog (Smog), with ozone and peroxyacyl nitrates (PAN) as important secondary products. Photochemical smog is caused by nitrogen oxide (NOx) and volatile organic compound (VOC) emissions. Therefore, the U.S. Environmental Protection Agency (EPA) decided, some 20 years ago, to combat photochemical smog in the U.S. by stringent reduction of VOC emissions and to a lesser extent, reduction of NOx; however, this policy did not produce the desired results. Natural emissions, especially of terpenes and isoprenes, proved to be so large that sufficient VOC concentrations were available in the atmosphere for oxidant production, despite efforts to limit anthropogenic sources.
Electric vehicles are sometimes vaunted for reducing air pollutants. However, firstly there are considerable emissions from the sources of the electricity. Secondly, the very high air pollution and carbon cost of manufacturing electric vehicle batteries negates any advantage of electric vehicles over complete life cycle comparison (e.g. manufacture, use and disposal). (Dai et al, 2019)
Emissions Acid Deposition and Oxidant Formation
| Table 1. Global, natural and anthropogenic emissions of SO2 and NOx | |||
|---|---|---|---|
| Emissions in Tg (1012)/year | SO2/IPCC | NOx/Graedel | Volatile Organic comp. |
| Biomass burning | 2.2 | 6 | 45 |
| Volcanoes | 9.3 | - | - |
| Lightning | - | 5 | - |
| Biogenic emissions from land areas | 1.0 | 15 | 350 (isoprenes) + 480 (terpenes) |
| Biogenic emissions from oceans | 24 | - | 27 |
| Industrial and utility activities | 76 | 22 | 45 |
| Solvents | - | - | 15 |
| Total natural emissions | 34 | 21 | 855 |
| Total anthropogenic emissions | 78 | 27 | 105 |
| Total emissions | 113 | 48 | 960 |
The anthropogenic sources of sulfur dioxide (SO2), (see Table 1) are much larger compared to natural sources; in fact, they exceeded natural sources as early as 1950. The natural emissions of NOx are, according to the estimate compiled by Graedel and Crutzen, in the same order of magnitude as anthropogenic emissions. Some NOx sources, especially natural ones, are quite uncertain, as is the contribution of natural sources to ambient VOC concentrations.
(It should be noted that the emissions due to biomass burning and from land areas are partly natural and partly anthropogenic. "IPCC" indicates data provided by Intergovernmental Panel on Climate Change. "Graedel" indicates data provided by Graedel and Crutzen).
Natural VOC emissions are predominant on a global scale, but near industrialized areas anthropogenic sources are the most important contributors to ambient concentrations.
A very recent overview of emissions of sulfur and nitrogen compounds is presented in Table 2. In this table the differences in emissions between Northern and Southern Hemisphere are highlighted, which makes clear that, in general, the North's contribution to emissions is much larger.
Sulfur dioxide emissions are declining in Europe and the U.S., while they remain constant or are increasing slightly in developing countries. Nitrous oxide emissions have stabilized or declined in developed countries, but are significantly increasing in developing countries. These trends suggest that acid deposition (a consequence of sulfur dioxide emissions) is being exchanged with exposure to increased oxidant concentrations.
Emissions: Global Problems
An extreme case is reactive chlorine (the main compound responsible for destroying stratospheric ozone, see Antarctic ozone hole), where at least 90% of the relevant emissions are anthropogenic. Chlorofluorocarbons (CFCs) were widely applied, beginning in 1945, until their use was banned through enactment of the Montreal Protocol in 1998. No natural sources of these compounds are known.important
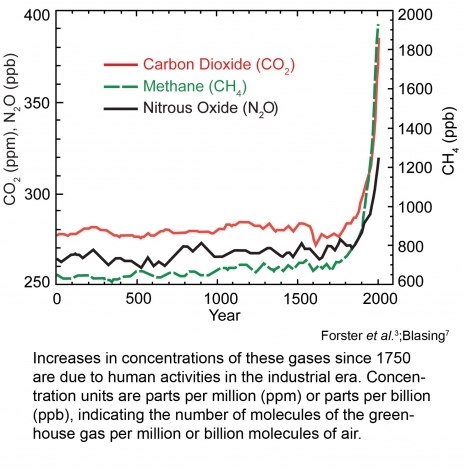
The atmospheric concentrations of the some of the greenhouse gases (CO2, CH4, CFCs and N2O) have been plotted from 1850 until present as a fraction of the present global ambient concentration. If it is assumed that ambient concentrations vary linearly with emissions (which is not wholly accurate), and that only natural emissions are responsible for concentrations before the year 1800, the ratio of natural versus anthropogenic sources varies significantly for the different [[greenhouse gas]es]. The ratio is highest for N2O and lowest for CFCs.
Aerosols
Aerosols, defined as solid or liquid particles which have a certain life time suspended in air (particle size varying from a few nanometers to several hundred micrometers, as in the case of dust particles), are either emitted as such (primary aerosol), or are formed by way of the transformations of pollutants such as sulfur dioxide, nitrogen oxides and ammonia into sulfates, nitrates and ammonium respectively (secondary aerosol). Many volatile organic compounds are converted to oxidized organic species with low volatility, thus becoming a component of ambient aerosol.
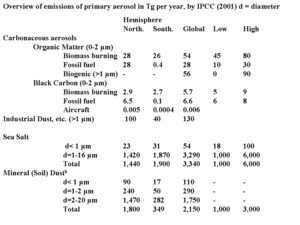
Table 4. Natural and human-made primary emissions of aerosols Ambient aerosol (Air pollution emissions) concentrations are partly of primary and partly of secondary nature. Sea salt particles caused by waves and dust swept up by wind are examples of primary aerosol. Primary aerosol emissions, especially dust and seasalt particles, constitute the largest contribution to total aerosol mass. An overview of primary emissions, both natural and human-made is given in Table 4.
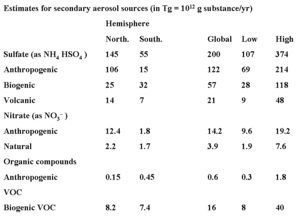
A large part of secondary aerosols, however, consists of sulfates and, depending on the specific conditions, nitrate and organic compounds, which are formed by atmospheric conversion with sulfur dioxide, nitrogen oxides and VOCs as precursors. An overview of the amount of secondary aerosol is found in Table 5. Near areas of high emissions (e.g., industrialized regions of Europe, the U.S. and Asia) the anthropogenic (human-made) emissions are more important than the natural.
Table 5. Secondary emissions of aerosols Sulfate still represents the largest fraction of anthropogenic contributions to ambient aerosols, but the present composition of aerosol reflects a shift in emissions, especially in developed countries in the last 10 years. For example, emissions of sulfur oxides have gone down due to measures to reduce acid deposition (Air pollution emissions) . Emissions from the transportation sector have been stabilized or even increased, notwithstanding the introduction of exhaust catalysts in cars, because some countries, e.g., the U.S. and Europe have seen an increase in the number of cars on the road as well as the total driven distances per car. As a result, there has been a shift from sulfate to nitrate in ambient aerosol.
Aerosols have a large impact on local pollution problems, but also contribute to regional environmental issues (e.g., acidification) and global environmental issues (e.g., the Antarctic ozone hole and climatic change (Causes of climate change)).
Sinks of air pollution
Some air pollutants have virtually no sink to remove the pollutant from the atmosphere. The chief chemical which has extensive natural sinks is carbon dioxide. While carbon dioxide is not really an air pollutant, it is a frequently cited emission, which has extensive natural sinks, especially in nurturing the growth of plant organisms, but also is extensively absorbed and sequestered in soils and oceans. A number of air pollutants have no sink mechanisms of note; for example, nitrogen trifluoride, one of the most powerful , gases has essentially no means of disintegration. Nitrogen trifluoride, a notable waste gas in the manufacture of solar panels, has a half life in the atmosphere of over 17000 years.References
- Qiang Dai, Jarod Kelly, Linda Gaines and Michael Wang (2019) Life Cycle Analysis of Lithium-Ion Batteries for Automotive Applications. Systems Assessment Group, Energy Systems Division, Argonne National Laboratory, DuPage County, Argonne, IL 60439, USA. Batteries 2019, 5(2), 48; https://doi.org/10.3390/batteries5020048
- Seiler W, Crutzen P (1980) Estimates of gross and net fluxes of carbon between the biosphere and atmosphere from biomass burning. Climatic Change 2: 207-247.
- "Slash-and-burn agriculture". Encyclopaedia Britannica.
- M.R.Beychok. 2005. Fundamentals Of Stack Gas Dispersion, 4th Edition, ISBN 0-9644588-0-2
- D.Bruce Turner. 1994. Workbook of atmospheric dispersion estimates: an introduction to dispersion modeling, 2nd Edition, CRC Press, ISBN 1-56670-023-X
- C. Michael Hogan and Harry Seidman. 1971. Air Quality and Acoustics Analysis of proposed I-66 through Arlington, Virginia, ESL Inc. Technical Document T1026, Sunnyvale, California In the matter of Arlington Coalition on Transportation v. Volpe, 458 F.2d 1323 (4th Cir.), 409 U.S. 1000
- C. Michael Hogan, Ballard George and Marc Papineau. ESL Inc. 1973. Air Quality, Noise and Energy Analysis of Tranportation Alternatives. Boston Transportation Planning Review. Boston, Massachusetts
- C. Michael Hogan, Marc Papineau Richard Venti. Federal Aviation Administration. 1973. Development of an air pollution line source model for analyzing aircraft operations. ESL Inc. U.S. FAA, Washington DC
- Jones, C.D. and Cox, P.M., 2005. On the significance of atmospheric CO2 growth rate anomalies in 2002–2003. Geophysical Research Letters, 32(14).
- Swift, Roger S. (November 2001). "Sequestration of Carbon by soil". Soil Science. 166 (11): 858–71. Bibcode:2001SoilS.166..858S. doi:10.1097/00010694-200111000-00010. S2CID 96820247.