Virus
A virus is a microscopic organism that can replicate only inside the cells of a host organism. Most viruses are so tiny they are only observable with at least a conventional optical microscope. Viruses infect all types of organisms, including animals and plants, as well as bacteria and archaea. Approximately 5000 different viruses have been described in detail at the current time, although it is known that there are millions of distinct types.[1] Viruses are found in virtually every ecosystem on Earth, and these minute life forms are thought to be the most abundant type of biological entity.[2] The study of viruses is known as virology, a specialty within the field of microbiology.
The common concept of viruses focuses on their role as pathogen. Actually, there are vast numbers of viral entities that are beneficial to individual species as well as providing ecosystem services. For example, a class of viruses known as bacteriophages can kill a spectrum of harmful bacteria, providing protection to humans as well as other biota.
Viruses are key in the carbon cycle; their role in ocean biochemistry includes microbological metabolic—including decomposition—processes. It is this decomposition that stimulates massive carbon dioxide respiration of marine flora. That respiration annililates effectively about three gigatons of carbon each year from the atmosphere. Significantly, viruses are being developed as tools for constructive modern medicine as well as the critical field of nanotechnology.
Unlike prions and viroids, viruses consist of two or three parts: a helical molecule, protein coat and sometimes a viral wrapper. All viruses have genesconstructed from either Deoxyribonucleic acid (DNA) or Ribonucleic acid (RNA)—long helical molecules that carry genetic information. All viruses have a protein coat that protects these genes, and someare wrapped in a viral envelope of fat that surrounds them when they are outside a cell. (Viroids do not have a protein coat and prions contain neither RNA nor DNA).
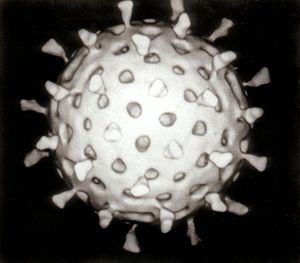
Viruses vary from simple helical and icosahedral shapes to more complex structures. Most viruses are approximately one hundred times smaller than an average bacterium. The origins of viruses in the evolutionary history of life are unclear. Some may have evolved from plasmids—fragments of DNA that can migrate between cells—while others may have evolved from bacteria. In evolution, viruses are an important means of horizontal gene transfer, which increases genetic diversity.
Contents
Lifeform or not?
Viruses have no ability to metabolize on their own, but depend upon a host organism for replication and manufacture of chemicals needed for such replication. Rybicki has characterized viruses as a form "at the edge of life".[3] Viruses are found in Modern taxonomy and that taxonomy considers viruses as a totally separate form of life from cellular organisms—and some would say that they are merely complex molecules with a protein coating and not a lifeform at all. Since viruses are capable of self replication, they are clearly some type of lifeform, and likely involved with the early evolutionary development of such other simple lifeforms as bacteria and protists.
Viruses differ, however, from the simpler autonomous replication of chemical crystals. This is since a virus can inherit a genetic mutation and is also subject to similar natural selection processes of cellular organisms. A virus cannot be labelled simply, therefore, as inanimate or lifeless. Here, we consider it a lifeform, but we adhere to current taxonomy and do not credit it with a parallel domain to other recognized cellular lifeforms.
Evolution
Although there is no detailed catalogue of the evolutionary relationships of viruses and hosts, certain gerneral characterisations can be made. In some such viral groups as poxviruses, papillomaviruses and tobamoviruses, molecular taxonomy aligns generally with the genetic relationships of their hosts.[4] This suggests that the affilations of those viral groups predate their present derivatives, and, in fact, that these three viral groups and their hosts likely co-evolved. There are clear examples where an otherwise genetically close group like the tobamoviruses include a genetiically outlying host; in particular, the tobamoviruses generally utilize plants of the Solanaceae family, but an orchid and a cactus virus can also be found in the group.
Recombination of genome parts of viruses poses a more vexing puzzle, since the events are virtually random pieces of an evolutionary chain. Retroviruses and luteoviruses are examples of viral groups where large numbers of recombinations have occurred to produce new organisms. Sometimes these produced genome splices occur naturally using fragments that are either viral or cellular in nature. In some cases the product is more of a re-arrangement of genomic parts—referred to as psuedo-recombination. The Western Equine Encephalovirus is a known example of this last category.
It is likely that viruses began host relationships with archaea and bacteria about two billion years ago; it has been suggested, however, that the proliferation of terrestrial vascular plants was the watershed event in evolution that enabled the explosion of numbers of viral organisms and pathways.[5]
Taxonomy
There are two complementary systems for viral taxonomy: the ICTV and Baltimore approaches. In the case of the ICTV taxonomy, there are five distinct orders: Caudovirales, Herpesvirales, Mononegavirales, Nidovirales, and Picornavirales. Within that hierarchy reside 82 families, 307 genera, 2083 species.[6]
David Baltimore devised an earlier system based on the method of viral messenger RNA synthesis.[7] The Baltimore scheme is founded on the mechanism of messenger RNA production. Although viruses must replicate mRNAs from their genomes to produce proteins and reproduce, distinctly different mechanisms are employed within each viral family. Viral genomes may be single ((ss) or double-stranded (ds), may be RNA or DNA based, and may optionally employ reverse transcriptase (RT); furthermore single strand RNA virus helices may be either sense (+) or antisense (−). These nuances divide viruses into seven Baltimore groups.
This Baltimore classification of scheme is centered around the concept of messenger RNA replication, since viruses generate messenger RNA from their genomic coding to produce proteins and thence replicate themselves. The resulting Baltimore groups are:
- I: dsDNA type (examples: Adenovirus, Herpesvirus, Poxvirus)
- II: ssDNA type (+)sense DNA (example: Parvovirus)
- III: dsRNA type (example: Reovirus)
- IV: (+)ssRNA type (+)sense RNA (examples: Picornavirus, Togavirus)
- V: (−)ssRNA type (−)sense RNA (examples: Orthomyxovirus, Rhabdovirus)
- VI: ssRNA-RT type (+)sense RNA with DNA intermediate to life-cycle (example: Retrovirus)
- VII: dsDNA-RT type (example: Hepadnavirus)
Morphology
The majority of viruses characteristically measure between 10 and 300 nonometers (nm), although certain filoviruses extend to a length of up to 1400nm, with a diameter of approximately 80nm.
A complete virus, known as a virion, consists of nucleic acid encased within an exterior protective coating of proteins termed a capsid—constructed from identical protein subunits called capsomers.
Very few viruses cannot be ovserved with a basic light microscope, but scanning and transmission electron microscopes can be employed to observe the viron or complete virus. To increase the contrast between viruses and the background, electron-dense staining is invoked; this technique involves solutions of heavy metal salts (e.g. tungsten), that can scatter impinging electrons from regions covered with the stain. When virions are coated with positive stain, fine detail is obscured, and negative stains (of the background only) are applied to complement the positive staining observations. There are four major distinct structural forms:
Helix
This group is characterized by single type of capsomer stacked around a core axis to form a helical structure, which may have a central cavity within the helix. This geometry results in rod-shaped or filamentous structures, which may be quite long, flexible and filamentose or abbreviateds and rigid. Most often the core genetic macro-molecule is single-stranded RNA is bound inside the protein helix by polar interactions between the negatively charged nucleic acid and effective positive charge at the protein surface. Tobacco mosaic virus is a prominent example of a helical virus.
Envelope
In some cases a cell membrane of the host is utilized for encasement of the virus; this may be either the external cell membrane or the nuclear membrane. These membranes become the outer lipid bilayer known as a viral envelope. The membrane is studded with proteins coded by the viral genome and host genome; the lipid membrane and any carbohydrates present derive exclusively from the host. The Influenza and HIV viruses use this strategy.
Icosahedral
Complex structures
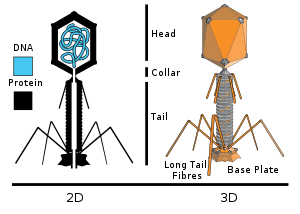
More complex viral structures may have a capsid that is neither purely helical, nor purely icosahedral, and that may possess such ancillary structures as protein tails or a complex outer wall. Some bacteriophages, including Enterobacteria phage T4 possess a complex structure of an icosahedral head bound to a helical tail, that may have a hexagon shaped base plate with protruding protein tail fibers. Such a tail performs as a molecular syringe, first attaching to the bacterial host, and then injecting the viral RNA or DNA into the host cell.
Replication
The virus, totally dependent upon its host for reproduction, manifests six essential stages in its life cycle:
- Attachment is the intermolecular binding between viral capsid proteins and receptors on the outer membrane of the host cell. The specificity of binding determines the host species and cell types that are receptive to viral infection. For example, HIV infects only human T cells, because the surface protein, interacts with CD4 and chemokine receptors on the surface of the T cell itself. This mechanism is thought to have evolved to discriminate in favor of those viruses that only infect cells in which they are capable of replication. Attachment to the outer host cell membrane may induce the viral-envelope protein to undergo changes that results in the fusion of vius and host cell membranes.
- Viral entry is the next step, wherein a virus penetrates the host cell wall. In the case of plant cells, the cell outer membrane is composed of cellulose, such that cell wall trauma must be usually precedent; however, certain plant viruses (for example, Tobacco mosaic virus) can pass from cell to cell through plamodesmata, or pore structures. Also, bacteria have significantly strong cell walls. Some viruses have evolved mechanisms that inject their DNA or RNA into a bacterium, with the viral capsid remaining outside.
- Uncoating is the step where viral enzymes degrade the virus capsid; sometimes host enzymes also play a role in this dissolution, that then exposes the viral genome to the interior of the host cell's chemical factory.
- Replication is the actual synthesis of (i) the virus messenger RNA (except for the case ofpositive sense RNA); (ii) synthesis of virus proteins; and (iii) assembly of replicated genomic material and subsequent protein binding.
- Post-translational modification of viral proteins sometimes transpires. For example, in the case of HIV, such a step (often termed maturation)happens once the virus has escaped out of the host cell.
- Lysis is the final step, in which the host cell dies through the act of its membrane being burst by the viral escape.In some cases the new virus genome becomes dormant in the host, only to come to life at a later time, when the activated virus subsequently lyses.
Pathology
There are numerous mechanisms by which viruses induce disease in an organism, chiefly depending on the virusl taxon. At the cellular level these mechanisms often include cell lysis, the breaking open and subsequent death of the cell. In multicellular organisms, if sufficient numbers of cells die, the whole organism may suffer gross metabolic disruption or even mortality. Although viruses may cause disruption of normal homeostasis, resulting in disease, in some cases viruses may simply reside inside an organism without significant apparent harm. An example, termed latency, is the ability of the herpes simplex virus, which includes cold sores, to remain in a dormant state within the human body.
Pathways of viral attack include respiratory intake, ingestion, body fluid contact and dermal contact. Each virus may have a different set of attack pathway characteristics, but prevention is difficult due to the microscopic size and ex vivo durability of viruses. The extremely small sub-cellular scale makes trapping of viruses by masks or filters virtually impossible.
Infections in humans
Human diseases caused by viruses include chickenpox, HIV, influenza, Marburg, ebola, Hanta, avian flu, cold sores and the common cold. The relative strength of viruses to induce disease is denoted by virulence.
Some viruses can induce chronic infection, such that a virus replicates over the entire remianing life of the host, in spite of the host's defence mechanisms. This syndrome is common in hepatitis B and C viral infections. Those with chronic infections are deemed to be carriers, and they are reservoirs of infectious virus as long as they live. For regional populations with a high carrier percentage, the disease is termed endemic.
Viral tranmission may be vertical (from mother to child) or horizontal (from individual to individual), with horizontal being the most common mechanism of viral propagation. Horizontal infection can occur via exchange of blood; exchange of body fluid by sexual activity; oral exchange of saliva; from contaminated food or water; respiration of viruses contained in aerosols;through animal or insect vectors such as mosquitoes. Each virus has a preferred method of transmission. The velocity of spread of viral disease correlates with a number of factors, human population density and sanitation being two of the most significant. There is evidence that many highly lethal viruses lie in biotic reservoirs in remote areas, and epidemics are sometimes triggered when humans encroach on natural areas that may have been isolated for long time frames; this theory has been advanced for ebola and HIV, that are thought to have resided in isolated African habiats for millennia.
Plant viruses
There are a large number viruses, that may manifest only as such superficial effects as fruit blemishing; however, crop yield reductions may result or even catasrophic loss of an entire plant population in a local area. Furthermore, control of these viruses may not be economically viable. In many cases viruses affecting vegetation may spread norizontally via third party organisms, termed vectors, that may be insects, fungi, nematodes or protozoans. Control of plant viruses usually consists of killing or removal of vectors that transmit the virus or removal of weed populations among crops that are secondary hosts Plant viruses are ineffective in infecting animals, since their replication is only functional in living plant cells.
Vegetative species exhibit elaborate defense mechanisms to ward off viral attack. One of the most effective defense mechanisms is the presence of resistance (R) genes. Each R gene confers resistance to a particular virus by triggering localized areas of cell death in proximity to the infected cell, which is often visible to the naked eye as large splotches. This phenomenon prevents the viral infection from spreading. An alternative defense is via RNA interference.
Bacteriophages
A bacteriophage is a virus that attacks a bacterium host, and is one of the most abundant organisms on our planet; they are found in soil, ocean water, aerosols and within animal intestinal tracts. For example as many as 900 million viruses may occur in one milliliter of seawater, situated in surface microbial matting;RNA synthesis.[8] the associated infection rate of marine bacteria may approach seventy percent.
The bacteriophage may inject its genome into the bacterial cell though its tail structure after planting itself with long tail fibers atop the host cell wall. Each bacteriophage is minuscule in relation to its host bacterium cell. Since many bacteria have become resistant to a spectrum of drugs, bacteriophages are viewed now as possible agents against certain drug-resistant bacterial pathogens.
Double stranded DNA tailed bacteriophages, comprise approximately 95 percent of the bacteriophages currently known. The major defense tactic bacteria employ is the production of enzymes that kill foreign DNA. These restriction endonucleases cut up the viral DNA thatis injected into the host cell.
Marine ecology and carbon cycling
Baceriophages, in particular, have a central function in marine ecology and carbon cycling. These organisms are extremely widespread in the world's oceans, sometimes occurring in concentrations as high as 900 million bacteriophages per milliliter. Secondly, they have a very rapid attack and replication cycle, being capable of attaching and injecting genomic material into a host bacterium in a matter of minutes, and acheiving genetic replication of new viruses on the order of 20 minutes. They are capable, therefore, of very rapid rates of multiplication in the marine enviornment.
Next, it is important to note that bacteriophages are highly correlated with concentrations of sewage. This is due to the presence of such bacteria present in untreated sewage as E. coli. In many world regions, large volumes of untreated sewage is discharged to the oceans. Without the ability of bacteriophages to systematically decompose the resulting high bacteria levels, not only would the bacterial concentrations be very high, but opportunity for enhanced carbon dioxide respiration at the atmosphere/ocean interface would be reduced. The outcome respiration rate for ocean absorption of atmospheric carbon is approximately three gigatons per annum,[9] which is a significant percentage of the fossil fuel combusition input to the atmosphere. Thus, further understanding of these viral processes may be key to grasping the world's carbon balance, and perhaps even making intelligent management decisions to avoid global greenhouse gas buildup.
References
R. A. Edwards and F. Rohwer. 2005. Viral metagenomics. Nat. Rev. Microbiol., vol 3, issue 6, pp 504–510 E. P. Rybicki. 1990. The classification of organisms at the edge of life, or problems with virus systematics. S Aft J Sci 86:182–186 Advances in Virus Research. 394 pages Adrian J. Gibbs, Charles H. Calisher and Fernando Garcia-Arenal. 1995. Molecular basis of virus evolution. 603 pages Karl Maramorosch, Frederick A. Murphy and Aaron J. Shatkin. 2003. Advances in Virus Research. 394 pages International Committee on Taxonomy of Viruses. 2008. Virus Taxonomy 2008. [Retrieved on May 11, 2010] David Baltimore. 1974. The strategy of RNA viruses. Harvey Lectures. vol 70, pp 57-74 K. E. Wommack and R. R. Colwell. 2000. Virioplankton: viruses in aquatic osystems Microbiol. Mol. Biol. Rev. vol. 64, issue 1, pp 69–114 C. A. Suttle. 2007. Marine viruses--major players in the global ecosystem. Nature Reviews. Microbiology. 5 (10):801–12