Concept
Version 12
Created by Boundless
Constant-Volume Calorimetry
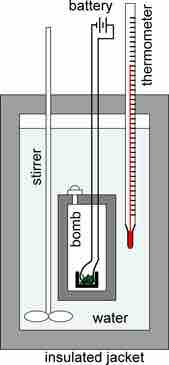
Bomb calorimeter
A schematic representation of a bomb calorimeter used for the measurement of heats of combustion. The weighed sample is placed in a crucible, which in turn is placed in the bomb. The sample is burned completely in oxygen under pressure. The sample is ignited by an iron wire ignition coil that glows when heated. The calorimeter is filled with fluid, usually water, and insulated by means of a jacket. The temperature of the water is measured with the thermometer. From the change in temperature, the heat of reaction can be calculated.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources:
"ChemicalPrinciplesFig2-4."
http://en.wikibooks.org/wiki/File:ChemicalPrinciplesFig2-4.jpg
Wikibooks
CC BY-SA 3.0.